News
Dunamis Medical announces FDA clearance of its PunchTac Anchor System for hip indications – Labral Repair/Reconstruction and gluteus medius repair
For Immediate Release
Contact: [email protected]
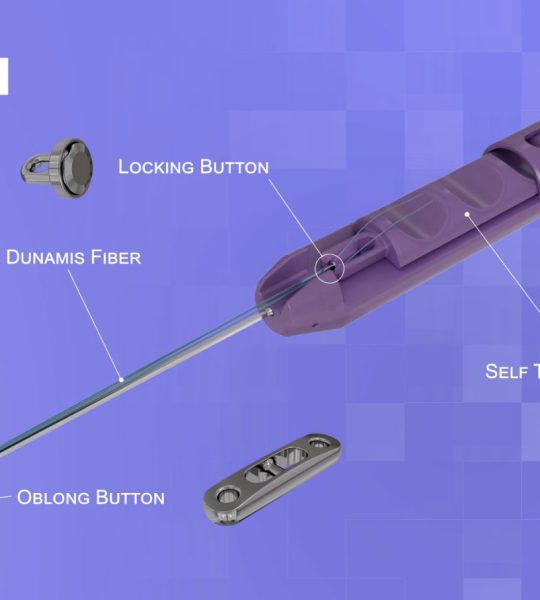
February 14, 2020 – Dunamis Medical® announces FDA clearance of its PunchTac™ Anchor System for hip indications – Labral Repair/Reconstruction and gluteus medius repair. Since the product launch, Dunamis’ PunchTac™ threaded and knotless anchors have been successfully implanted in patients.
Dunamis PunchTac™ anchors have shown to withstand axial impaction and have the necessary torsional strength when performing implantations in high-density bone.
Dunamis Medical® plans to begin an alpha launch April 2020 by collaborating with surgeons in multiple medical centers.
For additional information on approved indications, contraindications, warnings and potential adverse effects contact [email protected]
Latest news
Make a real difference.
Work with us
To request information about our company and products,
please start and complete the below form.
© Dunamis Medical, LLC 2017-